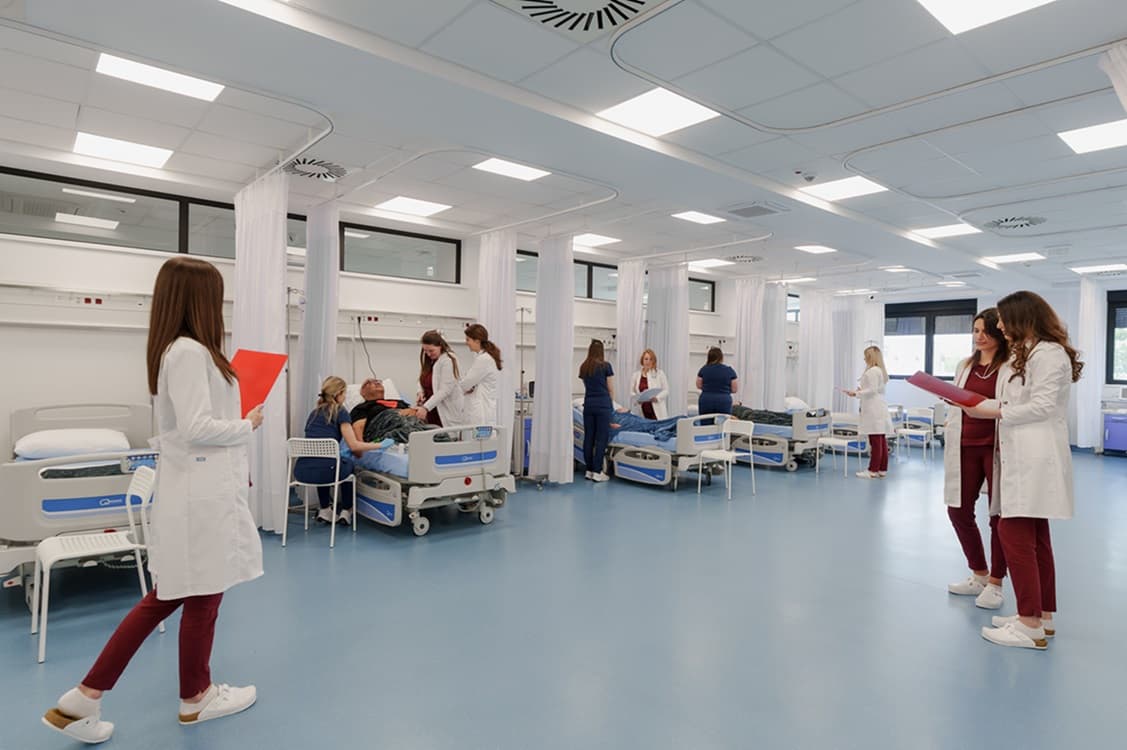
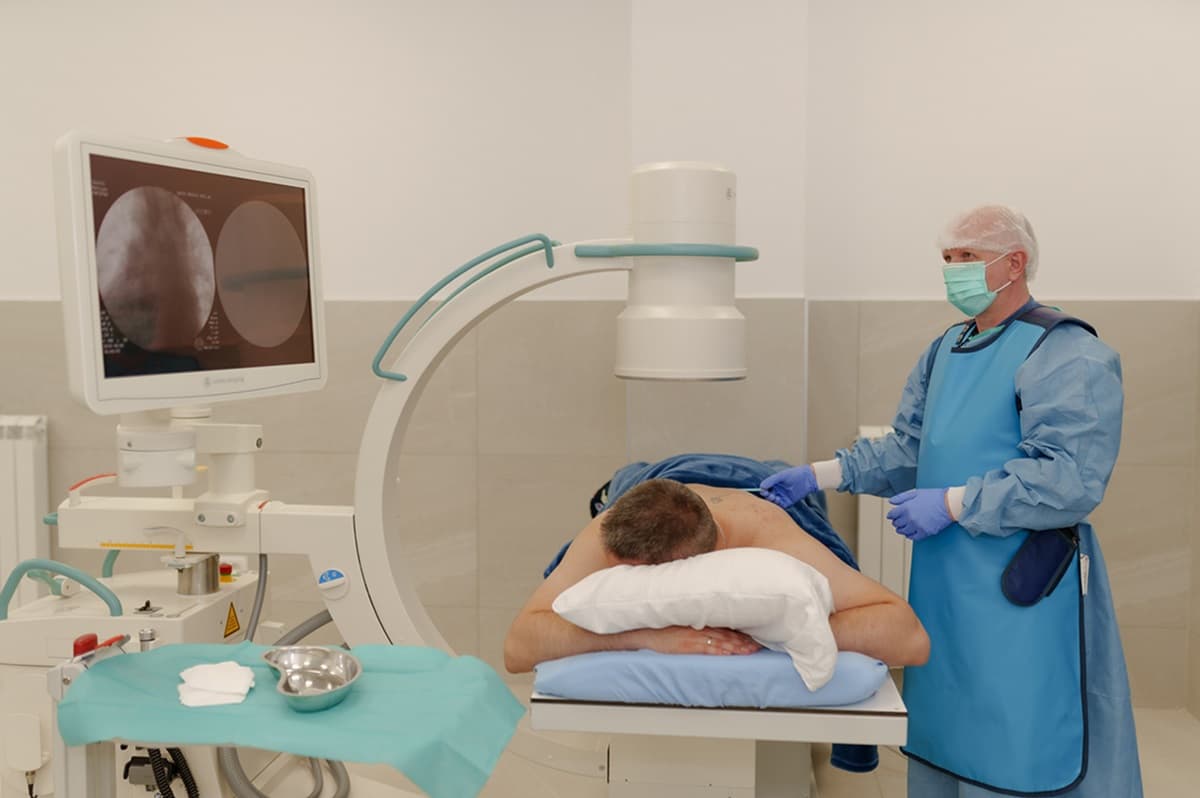
Parkinson’s disease (PD) is a progressive neurological condition, but for many people the first signs are surprisingly personal: a hand that trembles when reaching for a cup of coffee, steps that grow shorter, movements that become slower. These small changes can gradually reshape daily life long before the condition is officially diagnosed.
Medications often provide relief in the beginning, but over time many patients find that the benefits fade and conventional treatments cannot slow the disease’s progression.
Stem cell therapy for Parkinson’s disease offers a different path—focusing on regulating dopamine within neuronal pathways and reducing harmful inflammation in the brain, thereby supporting long-term neurological function.
In this article, we’ll look at how stem cell therapy for Parkinson’s disease can improve outcomes in addition to traditional treatments.
Understanding Parkinson’s Disease
Parkinson’s disease develops when certain changes in the brain start to affect how nerve cells work, especially those involved in movement.
What Causes Parkinson’s and How It Develops
Parkinson’s disease (PD) develops when nerve cells in a brain region called the substantia nigra gradually degenerate. These cells normally produce dopamine, a chemical messenger essential for smooth, coordinated movement.
The exact cause of this neurodegeneration remains unclear in most cases. Scientists suspect a combination of factors like aging, protein misfolding, environmental toxins, and oxidative stress may contribute. The disease progresses gradually—often starting years before noticeable symptoms appear.
How Parkinson’s Affects the Brain and Body
When dopamine drops, brain signaling becomes unbalanced, leading to motor symptoms like tremors, rigidity, slow movement, and poor balance. Parkinson’s also causes non-motor issues such as depression, anxiety, sleep problems, cognitive slowdown, and loss of smell.
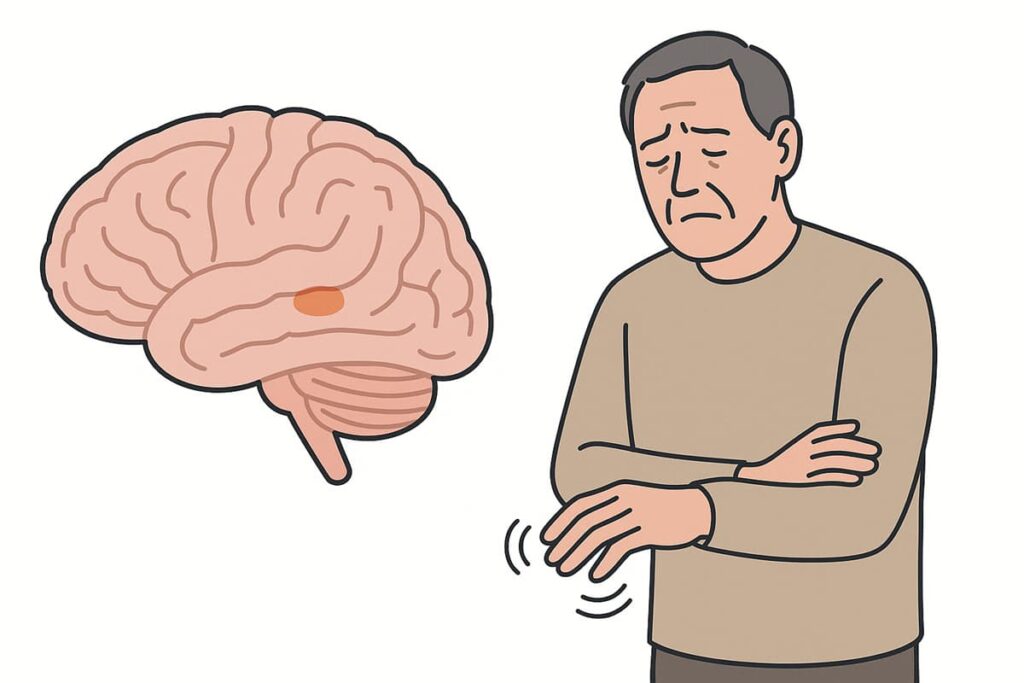
Early Symptoms, Diagnosis, and Disease Progression
Parkinson’s disease typically begins insidiously, with subtle symptoms that can be easy to overlook. Early indicators may include:
- Slight tremor in a finger or hand while at rest
- Smaller or cramped handwriting
- Ongoing constipation
- Reduced sense of smell
- Vivid dreams or physically acting out during sleep
- Softer voice or reduced volume
- Less facial expression
- Stooped or hunched posture
Diagnosis relies on clinical observation—there’s no single definitive test.
Parkinson’s is a progressive disease, meaning symptoms worsen over time. Progression rates vary, but generally the condition advances slowly over years or decades.
Conventional Treatments and Their Limits
The mainstay of Parkinson’s treatment is medication that boosts or mimics dopamine in the brain.
Standard Medications
The “gold standard” of long-term Parkinson’s management is the drug Levodopa, which aids dopamine delivery to the brain. Levodopa is absorbed and converted into dopamine, which can reduce symptoms—especially in the early or moderate stages of the disease. There are also dopamine agonists, which aren’t dopamine but act on dopamine receptors to simulate its effect.
While these medications do not stop the disease, they can improve day-to-day functioning.
Why Drugs Lose Effectiveness Over Time
Over the years, many patients find that Parkinson’s medications become less predictably effective. This is partly because Parkinson’s disease continues to progress—more dopamine neurons are lost, so there is less natural dopamine for medications to work with.
As the disease advances, medications alone often cannot fully eliminate tremors, stiffness, or balance issues.
The Need for Regenerative Approaches
Medication is a symptomatic band-aid on a progressive degenerative process. Stem cells for Parkinson’s disease are a regenerative complementary therapy that aims to address neuronal loss by creating a more neuron-friendly environment and supporting potential neuronal growth.
Types of Stem Cells Used in Parkinson’s Treatment
Potentially effective treatments for Parkinson’s disease include a variety of stem cell types.
Mesenchymal Stem Cells for Parkinson’s Disease
Mesenchymal stem cells (MSCs) are the most commonly used type of stem cells in Parkinson’s therapy protocols. Taken from bone marrow, fat, or umbilical cord tissue—either from the patient or a donor—they help reduce inflammation, regulate immune activity, and support nerve cell survival. They are known for their versatility and safety profile.
Some regenerative medicine clinics, like Swiss Medica, also use more specific neural stem cells to help restore dopamine-producing neurons and improve movement.
Neural stem cells can protect the brain, modulate inflammation, and potentially replace lost dopamine-producing neurons. Learn more about how they work and how they’re used for chronic conditions in our dedicated article.
Read moreStem Cell Transplant for Parkinson’s Disease: Infusion, Not Surgery
At Swiss Medica, a stem cell therapy clinic in Serbia, the term “stem cell transplant” refers to a minimally invasive infusion of stem cells, not a surgical procedure. MSCs are typically introduced into the body through an intravenous (IV) drip or a lumbar puncture injection into the cerebrospinal fluid (intrathecal administration). No general anesthesia is required.
Supportive Biological Therapies
Stem cells often work best in combination with other supportive therapies:
- PRP (platelet-rich plasma)
Uses the patient’s own blood platelets to release growth factors that aid healing and reduce inflammation. When paired with stem cells, it can boost repair signals in the brain. - Exosome therapy
Delivers tiny cell-free particles from stem cells that act faster than MSCs because they already contain active molecules like growth factors and cytokines. Unlike stem cells, they do not require time to adapt. - Macrophages
These immune cells may support repair by reducing inflammation and clearing damaged cells, potentially slowing disease progression. - Antioxidant protocols
Focus on reducing oxidative stress and supporting brain regeneration. This may involve IMR therapy (Intracellular Metabolism Recovery Therapy), which supports cellular metabolic processes by providing important nutrients like vitamins, amino acids, and various antioxidants. It can be combined with ultrasound or electrotherapy to improve cellular health and promote a better recovery environment.
At Swiss Medica, we tailor IMR therapy to each case and condition severity. Learn how it works and what results patients experience.
Read moreHow Stem Cell Therapy Works for Parkinson’s Disease
Stem cell work is not a miracle; it is based on specific mechanisms that help the body recover.
How Can Stem Cells Treat Parkinson’s Disease?
Stem cells have several properties that make them suited to treat Parkinson’s disease in a multi-faceted way.
1. Regenerating and Protecting Dopamine-Producing Neurons
Certain types of stem cells for Parkinson’s work in different ways:
- MSCs support existing neurons by releasing growth factors that help neurons survive longer, function better, and form new connections. MSCs also promote new blood vessel growth to improve blood supply—that may enhance neuron health and slow disease progression.
- Neural stem cells can potentially replace dopamine-producing neurons lost in Parkinson’s by differentiating into dopaminergic cells, helping restore dopamine levels and improve motor function.
2. Restoring Neural Communication Pathways
Both MSCs and neural stem cells can promote neural repair and encourage the brain’s own stem cells to form new connections—this is known as neuroplasticity, the brain’s capacity to rewire itself. By stimulating neurogenesis—the process of forming new neurons—stem cell transplants for Parkinson’s disease help create new neurons and connections.
3. Reducing Oxidative Stress
Parkinson’s disease is not only about losing neurons; it’s also characterized by a harmful cellular environment. Mesenchymal stem cells for Parkinson’s disease help counteract these destructive processes. They secrete antioxidants and anti-apoptotic factors that reduce oxidative stress in the brain, essentially neutralizing some of the “chemical fires” that burn neurons out.
Can Parkinson’s Disease Be Cured with Stem Cells?
Neuroregeneration therapy with stem cells is not a magic cure for Parkinson’s disease, but it can be a powerful tool to control the condition. Instead of thinking in terms of an alternative for a Parkinson’s cure, it’s more accurate to view stem cell therapy as a regenerative and immunomodulatory therapy that protects existing neurons, improves motor function, and slows the progression of symptoms.
Get a free online consultation
Want to know if stem cell therapy could help in your case?
Speak with a medical advisor for a free, no-obligation online consultation and receive honest answers about the outcomes.

Medical Advisor, Swiss Medica doctor
Expected Improvements and Long-Term Outcomes
While every individual responds differently, there are several common areas of improvement that have been observed:
- Tremor improvement and reduced rigidity. Tremors often become milder, and muscles feel less stiff or tense.
- Better balance and coordination. Patients may walk more steadily, regain arm swing, and feel more stable on stairs or uneven surfaces.
- Improved mood, sleep, and focus. Many feel less anxious or depressed, sleep more deeply, and think more clearly.
- More energy. Less fatigue means more activity—patients often return to hobbies, exercise, or social life.
- Greater independence and daily function. Everyday tasks like dressing or cooking become easier and quicker to manage.
Comparing Stem Cell Therapy with Other Parkinson’s Treatments
Rather than an “either/or” choice, stem cell treatment for Parkinson’s is typically complementary, adding a regenerative dimension to a patient’s care plan.
| Treatment | Main Approach | Key Benefits | Limitations | How Can Stem Cells Treat Parkinson’s Disease in Addition |
| Drug Therapy (Levodopa, dopamine agonists) | Increases or mimics dopamine levels | Fast symptom relief, especially early on | Does not repair neurons or slow disease progression | Stem cells may reduce need for meds over time but don’t replace them immediately |
| Surgery (Deep Brain Stimulation) | Electrical impulses regulate motor signals | Effective for tremor and rigidity; long-term option | Invasive; doesn’t treat non-motor symptoms or neuron health | Can be combined with stem cells; stem cells aim to repair circuits rather than bypass them |
| Rehabilitation + Nutrition (Physiotherapy, speech therapy, diet) | Improves mobility, coordination, daily function | Supports independence and slows decline | Needs consistent effort; limited biological effect on disease | Stem cells can boost energy and recovery, making rehab more effective; lifestyle helps support cell therapy outcomes |
Clinical Evidence and Observed Results
Scientific studies on stem cell therapy for Parkinson’s disease are rapidly progressing, with promising results.
What Research Shows about Stem Cell Therapy for Parkinson’s
Parkinson’s clinical trials from 2022 demonstrated that various sources of MSCs are safe and efficient:
- Bone marrow MSCs: patients experiencing better mobility, improved facial expression, fewer freezing episodes, lower Unified Parkinson’s Disease Rating Scale (UPDRS) scores, and in some cases reduced medication use.
- Umbilical cord MSCs: patients showed steady improvements in motor symptoms within 1–3 months, often with better energy, mood, and daily function, in both autologous and donor-derived treatments.
- Adipose-derived MSCs: patients felt consistent symptom improvement, with all patients showing lower UPDRS scores.
In 2023, a meta-analysis reported that cell-based therapies with 210 patients had preliminary beneficial effects on Parkinson’s patients’ motor function.
Success Rate of Stem Cell Therapy for Parkinson’s Disease at Swiss Medica
Swiss Medica has been at the forefront of regenerative medicine since 2011, and we monitor outcomes to ensure ongoing effectiveness. 80% of patients respond with noticeable improvements after undergoing their stem cell treatment program. We define stem cell transplant success rate in terms of patient-reported improvement in symptoms and quality of life measures.
Real Patient Success Stories
Here are two real stories highlighting outcomes for Parkinson’s patients at Swiss Medica.
— Patient from Sunshine Coast, Australia
“Before treatment my walking was bad, my left leg dragged, and I couldn’t even use a knife and fork—my wife had to cut my food. After each round of stem cells, I’ve improved: my hands recovered, my anxiety dropped, my concentration returned, and I can work full days again. This therapy has given me back a normal life.”
— A family from the Czech Republic
“After only a few days of treatment and physiotherapy at Swiss Medica, my father showed noticeable improvements—walking straighter, with better posture and more independence—after previously requiring near 24/7 assistance due to severe mobility decline.”
You can find more video testimonials about results of stem cell therapy for Parkinson’s disease in the US and other countries on our YouTube.
The Swiss Medica Parkinson’s Treatment Process
Swiss Medica has a structured, step-by-step process for treating Parkinson’s disease with stem cells. Here’s an overview of what a patient can expect:
Step 1. Comprehensive Diagnostics and Evaluation
Before any treatment begins, we conduct a thorough evaluation. Our specialists in regenerative medicine review your medical records, tests, and imaging to assess eligibility and overall health. Then we develop a personalized treatment plan that may include stem cell therapy, complementary therapies, and supportive care, which typically consists of 3–9 days of in-clinic treatment followed by up to 5 weeks of at-home support.
Step 2. Cell Preparation and Laboratory Quality Control
Once your treatment plan is confirmed, we prepare your stem cells in our own laboratory. Whether they come from your own tissues or a donor, every batch is thoroughly tested to ensure it’s safe, pure, and effective. During this process, we also produce exosomes and secretome from the cultivated cells.
Step 3. Stem Cell Administration and Neurological Monitoring
We deliver stem cells through a painless procedure—either an IV drip, an intrathecal injection, or a combination, depending on your needs. It usually takes under an hour, targets several symptom areas, and is closely monitored by doctors; most patients feel well afterward, with only occasional mild headache or fatigue.
Step 4. Rehabilitation, Physiotherapy, and Follow-Up
Based on your personalized treatment plan, we may include supportive therapies such as physiotherapy, IMR, occupational therapy, and speech therapy during your stay. After you return home, our doctors follow up at 3–6 months to check your progress.
Why Choose Swiss Medica
Swiss Medica is frequently cited by patients as one of the best stem cell therapy clinics in the world for the following reasons:
- Extensive experience in neurology—over 14 years specializing in regenerative treatments for Parkinson’s and other neurological and chronic conditions, with 10,000+ patients treated since 2011.
- A modern hospital built in 2024 with 24-hour medical staff and advanced rehab equipment; private, comfortable rooms that feel like home; nutritious meals; and its own green space to walk and relax.
- In-house GMP laboratory—we process and test stem cells on-site under EU GMP Grade A standards, ensuring high safety, purity, and viability. Every cell batch is checked for contamination and function.
- Personalized protocols and long-term support. From the moment we meet a patient, we develop a completely personalized treatment plan: from appropriate dosage and additional procedures to lodging and follow-up.
- Accessible location. Serbia is considered one of the best countries for stem cell treatment and is easy to reach, with most international flights taking just 3–6 hours.
Swiss Medica is a modern hospital with its own laboratory, well-equipped procedure rooms, comfortable accommodations, and nutritious meals.
Cost of Stem Cell Parkinson’s Treatment
The total stem cell treatment cost depends on:
- Cell source: your own vs. donor
- Number of cell doses and total cell count
- Length of stay: short vs. extended programs
- Additional therapies: rehab, oxygen therapy, physiotherapy, etc.
How Much Does Stem Cell Parkinson’s Treatment Cost?
At Swiss Medica, the cost ranges from €7,000 to €31,000*, depending on treatment complexity. Pricing is personalized and includes all medical procedures, stem cell sourcing, supportive therapies, meals, and pickup at the airport.
| Location | Estimated Cost of Stem Cells for Parkinson’s treatment | Notes |
| Serbia (Swiss Medica) | €7,000–€31,000* | Includes full program with medical care, stem cell prep, and support therapies |
| United States/ UK / Western Europe | $20,000–$50,000+ | Often limited to clinical trials; higher due to regulation and overhead |
While stem cell therapy for Parkinson’s disease in the US is often significantly more expensive—sometimes exceeding $50,000—Swiss Medica offers comparable safety, quality, and personalization at more accessible pricing.
*Prices are indicative, based on January 2025, and may vary with condition severity and cell quantity required.
Getting Started with Consultation
If you’re considering stem cell therapy for Parkinson’s, the first step is simple: reach out to Swiss Medica for a free consultation. You’ll be asked to share your medical history—diagnosis details, current symptoms, scans, and any test results you have.
If you are a suitable candidate, you will receive a personalized treatment plan with a clear outline of procedures, pricing, and next steps—with no obligation to commit.
Contact us
Contact Swiss Medica and get a free online consultation to learn if stem cell therapy could support your recovery.

Medical Advisor, Swiss Medica doctor
FAQ
1. How long do the results last?
Stem cells stay active for about 7–8 months, and many patients continue to feel better for up to 1.5 years after treatment, depending on their individual case.
2. What are the risks of stem cell treatment for Parkinson’s?
Most people tolerate the treatment well. Some experience fatigue or get a mild headache or fever for a day or two. Serious side effects are rare when the therapy is done properly in a medical setting.
3. How can stem cells treat Parkinson’s disease?
Stem cells may help ease symptoms and create a healthier environment in the brain, potentially slowing neuron loss and disease progression.
4. Will I still need my Parkinson’s medication?
Yes. Stem cell therapy is always used in conjunction with traditional treatments and may help you recover faster but will not cure you completely. Stem cells cannot replace standard treatments, but if therapy is effective, your doctor may gradually reduce your dosage.
List of References:
Jin Young Shin, Phil Hyu Lee, Mesenchymal stem cells modulate misfolded α-synuclein in parkinsonian disorders: A multitarget disease-modifying strategy, Stem Cell Research, Volume 47, 2020, https://doi.org/10.1016/j.scr.2020.101908
Wang, Y., Yi, H. & Song, Y. The safety of MSC therapy over the past 15 years: a meta-analysis. Stem Cell Res Ther 12, 545 (2021). https://doi.org/10.1186/s13287-021-02609-x
Stoker TB. Stem Cell Treatments for Parkinson’s Disease. In: Stoker TB, Greenland JC, editors. Parkinson’s Disease: Pathogenesis and Clinical Aspects [Internet]. Brisbane (AU): Codon Publications; 2018 Dec 21. Chapter 9. Available from: https://www.ncbi.nlm.nih.gov/books/NBK536728/
Wang, F., Sun, Z., Peng, D. et al. Cell-therapy for Parkinson’s disease: a systematic review and meta-analysis. J Transl Med 21, 601 (2023). https://doi.org/10.1186/s12967-023-04484-x
Heris, R.M., Shirvaliloo, M., Abbaspour-Aghdam, S. et al. The potential use of mesenchymal stem cells and their exosomes in Parkinson’s disease treatment. Stem Cell Res Ther 13, 371 (2022). https://doi.org/10.1186/s13287-022-03050-4
Oz T, Kaushik A, Kujawska M. Neural stem cells for Parkinson’s disease management: Challenges, nanobased support, and prospects. World J Stem Cells. 2023 Jul 26;15(7):687-700. doi.org/10.4252/wjsc.v15.i7.687
Medical Advisor, Swiss Medica doctor