Stem cell therapy for autism is gaining attention in the medical world for its ability to activate the body’s own healing processes. A recent clinical review of 461 children with autism spectrum disorder (ASD) reported notable improvements with no serious side effects, particularly in social interaction, motor skills, and behavioral development. From better eye contact to improved communication, the results are encouraging.
Could stem cell treatments for autism become a new standard alongside traditional behavioral strategies? Let’s look at why stem cells are gaining popularity and how they’re helping real children with autism—through science and lived experiences.
Why Do Parents Choose Stem Cell Therapy for Their Children with ASD?
For many parents, traditional autism treatments only go so far. They’re left searching for something that might unlock new progress for their child.
Stem cell therapy is always surrounded by both hope and confusion. While the potential is real, so are the risks of choosing unproven clinics. The truth is, the effectiveness of treatment largely depends on the quality of the therapy, the source of the cell, strict safety standards, and the individual needs of each child.
At Swiss Medica, we’ve treated over 3,000 children with ASD using advanced, personalized protocols—and we’ve seen firsthand why so many families are turning to stem cells to treat autism.
Reason #1: Real Progress They Can See in Their Child
In our experience, around 80% of families report noticeable improvements in their child within 3–6 months of therapy, especially in areas like:
- Communication: Using more words, making clearer gestures, responding when their name is called.
- Social skills: Increased eye contact, initiating play, showing interest in others.
- Behavior: Fewer meltdowns, less repetitive actions.
- Daily living: Gains in self-care, improved sleep, better hygiene habits.
- Physical health: Improved digestion, appetite, and comfort.
Each small step forward—be it a new word, a calm meal at a restaurant, or a hug from a once-distant child—represents a huge win for the family.
“We tried everything for years—speech therapy, reflex integration, horseback riding. There were almost no improvements. But after stem cell treatment, something changed.”
“His understanding got better. His focus improved. And now, he doesn’t just toss a ball—he wants to play. His play is meaningful. That means everything to me.”
Reason #2: Multiple International Studies Have Reported on the Safety and Effectiveness of Stem Cells
In autism, certain brain connections do not form or function properly. One of the underlying causes is chronic neuroinflammation, which interferes with normal brain function by limiting oxygen flow, nutrient delivery, and overall neural connectivity. This disruption can be described as an internal “storm” in the brain and make it harder for a child to communicate, learn, or form relationships.
That’s where stem cell therapy enters the picture.
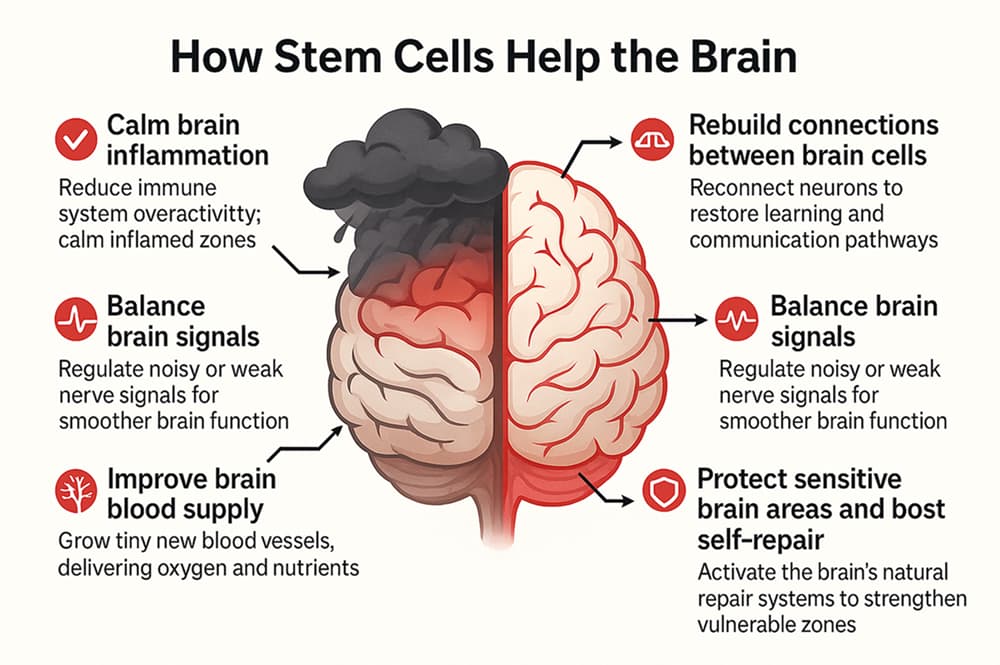
Over the past few decades, scientists have been exploring stem cell therapy as a way to calm this inflammation and restore balance in the brain. Clinical trials involving mesenchymal stem cells (MSCs)—the type we use at Swiss Medica—have shown that this therapy can help autistic children without causing any serious side effects.
Here’s how MSCs may support children with ASD, according to published research:
- Reducing brain inflammation
- Increasing activity of regulatory immune cells
- Improving blood flow and oxygen supply
- Releasing neurotrophic factors that support neurons, boosting brain communication, social cue processing, and learning ability
“Despite positive research findings, we do not view stem cell therapy as a universal solution for autism spectrum disorders. Our approach is based on leveraging the regenerative and immunomodulatory properties of MSCs to reduce neuroinflammation and support neuroplasticity, thereby creating a more favorable environment for the child’s ongoing development.
In the majority of cases, we observe measurable improvements—whether in the form of new skills, increased verbal output, or reduced anxiety and sensory dysregulation.”
Alexander Hitchregenerative specialist at Swiss Medica
Why Families Trust Swiss Medica for Autism Stem Cell Treatment
Choosing the right clinic for stem cell therapy for autism with a safe, child-friendly environment is just as important as the therapy itself. We know that stress can overshadow even the most promising therapy. That’s why parents from the U.S., U.K., India, and beyond tell us the same thing: they chose Swiss Medica for our comprehensive and child-centered approach.
From your first conversation with us to every little step forward your child takes, our support is always by your side.
Stem Cell Treatment for Autism at Swiss Medica: What’s The Difference
We follow four fundamental principles that enable us to offer one of the best stem cell treatments for autism in the world.
1. Scientifically Advanced, Carefully Delivered Stem Cell Products and Procedures
At Swiss Medica, we use the latest science to guide every step of treatment. We offer MSC therapy for ASD, which includes more than just injections. Here are the components of our therapies:
- High-quality stem cells: We use MSCs derived from ethically sourced umbilical cord and placenta tissue. Thanks to our in-house lab, every child receives fresh, highly viable cells prepared under strict, sterile conditions.
- Beyond just stem cells: To enhance the effects of MSC therapy, our program can also include supportive cell-based therapies:
- Exosomes are tiny messenger particles released by stem cells; they act quickly to reduce inflammation and support neurons, often providing a “jump start” even before the stem cells themselves become fully active.
- Macrophages are specialized immune cells—they help calm inflammation, clear out cellular debris, and create a healthier environment in the brain. This can potentially improve focus and sensory processing for the child.
- Personalized protocols: No two children with autism are the same, so we never use a one-size-fits-all treatment plan. Our medical team carefully adjusts the number of stem cell infusions, the dosing, and any supplemental therapies to your child’s specific needs. Factors like the child’s weight, the severity of ASD symptoms, medical history, and even previous treatment responses are all taken into account. We prepare a plan that best fits your child, rather than asking your child to fit a generic protocol.
- Non-invasive autism treatment options, child-friendly procedures: We make the treatment process as easy as possible for the child. No general anesthesia is required. Stem cells are administered through a simple IV drip or a quick injection, and exosome therapy is given via a painless nasal spray or inhalation. If a child is very nervous or sensitive, we can use a mild sedative to help them stay calm, but in most cases this isn’t necessary. The majority of children remain comfortable in our care—our procedure rooms are bright and soothing, with no harsh noises or glaring lights.
- Experienced medical team: Our doctors have over 15 years of experience in stem cell transplants for autism, having treated hundreds of cases and published research. This breadth of knowledge allows us to deal with psychological nuances like sensory issues or anxiety during therapy with stem cells for kids with autism.

2. Flexible Program Options for Every Child
Every family’s needs and schedules differ. That’s why we offer flexible treatment programs for autism, which vary in length and intensity to meet your child’s needs and the specifics of their condition.
| 3-Day | Short stay with one MSC infusion and exosome therapy, plus take-home vials for continued use. |
| 5-Day | Includes one MSC injection, multiple exosome sessions, and closer monitoring over nearly a week. This gives a bit more time for supportive therapies and observation while still being manageable for school schedules or working parents. |
| 9-Day | Intensive stem cell therapy for autism, which includes multiple MSC doses, advanced immune therapies (like macrophage injections if suitable), and daily supportive treatments (such as physiotherapy or occupational therapy sessions). |
| 2+3 Day | Specially designed for children under 3, split into two short visits a few months apart for your comfort and safety. This way, the child and parents aren’t overwhelmed by a long visit. |
Get a free online consultation
Not sure which program is best for your child? You don’t have to decide alone. Talk to our medical advisor about your child’s condition, and we’ll help figure out the most suitable plan. We provide a free online consultation where you can learn more about each option and get advice on what would benefit your child.

Medical Advisor, Swiss Medica doctor
3. A Place Where Families Feel Safe
Any medical treatment—and autism stem cell treatment—can be intimidating for a child with autism, so we go the extra mile to create a stress-free experience. Here are some of the ways we make our clinic family-friendly and comfortable for kids:
- Parent involvement: At Swiss Medica, parents are not just allowed but welcomed to stay with their child throughout the treatment process. You can be by your child’s side during all procedures. We believe a parent’s presence is incredibly reassuring to a child.
- Stress-free travel experience: We know you might be coming a long way from home, and this whole experience could feel daunting. From the day you arrive, we assign you a personal assistant/translator who will be by your side to handle any practical issues and answer questions. We can handle airport transfers and local transportation so you don’t have to worry about them. The result is that you can genuinely relax and focus on your child’s therapy and progress.
- Home-like setting & personalized comfort: Cozy rooms designed specifically for families with children, with a quiet atmosphere and sensory-friendly spaces. All of our treatment plans come with daily nutritious meals, with options to accommodate special diets, such as gluten-free or casein-free.
- All-inclusive family support and on-site amenities: We take care of the little things so you can focus on your child. There are indoor and outdoor play areas and safe sensory rooms where your child can unwind or have fun between treatments.
- Specially trained, compassionate staff: Every member of our team, from doctors and nurses to support staff, is experienced in working with children on the autism spectrum. We train our staff to approach each child in a way that respects their individual communication style.
Swiss Medica is more than a hospital—it offers on-site family apartments, nutritious meals, and play areas for children.
4. You’re Not Just a Patient—You’re Family
Our top priority is to build a trusting relationship with you and your child. We believe that follow-up and ongoing support are just as important as treatment, because autism is a lifelong journey. Here’s what you can expect from our patient-centered approach:
- We listen first: Every child with autism has their own story. We listen to your child’s medical history, but also to your insights as a parent—your child’s likes and dislikes, what triggers to avoid, what goals you hope to achieve. This allows us to tailor the treatment.
- Compassionate care: We make sure you feel seen, heard, and respected at every step, with no rushed appointments or obligations. During your stay, we check in 24/7 to see how you and your child are doing, not just physically but emotionally as well.
- Long-term support: After the treatment, we stay in close contact to monitor your child’s progress. Typically, we schedule a check-up call or video consultation about 3 months after the therapy to discuss any improvements or challenges. You’ll also have direct contact info for our doctors and medical staff, so you can reach out with questions at any time.
- Community connection: Many families stay in touch, share milestones, and return for future care.
To see for yourself what a day at Swiss Medica is like, we invite you to watch a brief video tour on our YouTube channel.
Who Is a Good Candidate for Autism Stem Cell Treatment?
Stem cell therapy for autism can be promising, but it’s not a miracle cure or a fit for every single case. We have specific criteria to ensure that we only treat children who are likely to benefit and for whom the procedure will be safe. These are the main factors we assess before recommending stem cells in autism spectrum disorder.
Age Groups, ASD Stages, and Other Key Evaluation Criteria
| Age Range | The age limit for stem cell therapy for autism is mainly 3–18 years, with the best response typically in children 3–7 years old. |
| Weight | To ensure safety, a child should weigh at least 15 kg (33 lbs). |
| Comorbid Conditions | MSC therapy for ASD is especially beneficial for children with gastrointestinal issues (e.g., chronic constipation or diarrhea) or sleep disorders (insomnia, irregular sleep cycles). |
| No serious mental diseases | No accompanying psychiatric conditions, such as schizophrenia. |
| ASD Severity Levels | Mild, moderate, and severe cases are all considered; earlier stages respond faster, but even severe cases can improve (e.g., calmer behavior, better sleep). |
| Active parental Involvement | Autism stem cell treatment can provide a boost in neurodevelopment, but to truly maximize and maintain the gains, a child needs continuous support and learning opportunities. We especially encourage that parents are actively working with the child in ongoing therapies or educational programs—such as speech therapy, occupational therapy, ABA therapy, or special education. |
Sometimes we’re asked if there is an age limit for stem cell therapy for autism in adulthood. Unfortunately, the results for adults are often less impressive. The younger the patient, the more the brain can potentially rewire in response to therapy.
Get a free online consultation
If you’re unsure whether stem cell therapy could help your child, book a free online consultation. Our doctors will review your child’s medical history to assess eligibility, safety, and potential benefits—with no obligation.

Medical Advisor, Swiss Medica doctor
How the Process Works—Step by Step
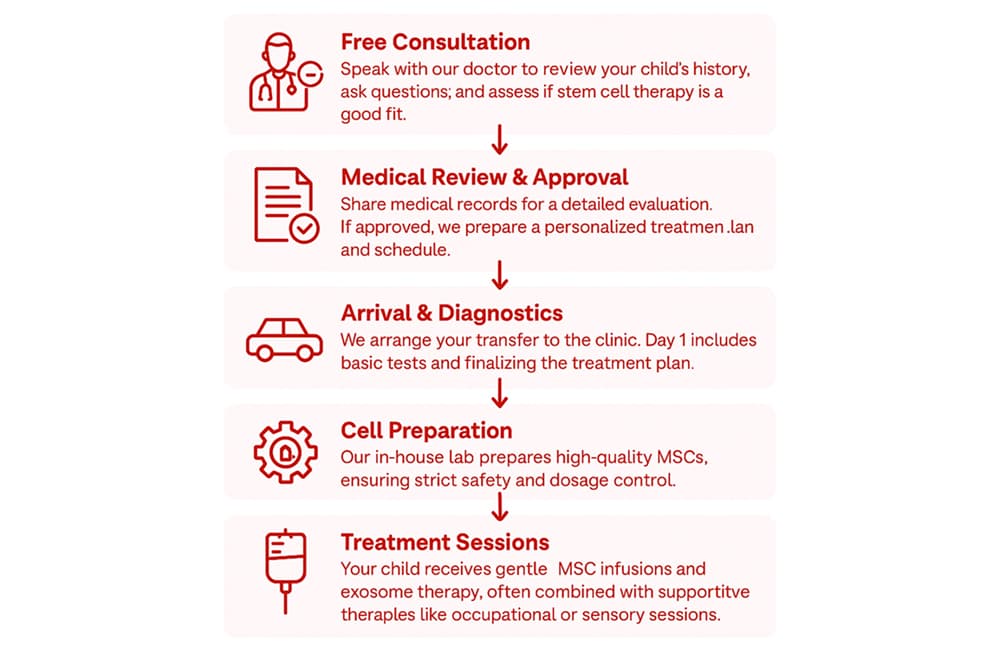
Undergoing stem cell treatment for autism might sound complex, but we make the process simple for our patients. Here’s a step-by-step overview of how it works, from your initial inquiry all the way through post-treatment follow-up:
- Free consultation: Speak with our medical advisor to review your child’s history, ask questions, and assess if stem cells are beneficial in your child’s case.
- Medical review & approval: After the first consultation, we will ask you to send over your child’s relevant medical records (recent doctor reports, test results, etc.). Our medical team prepares a personalized treatment plan and schedule for your child.
- Arrival & diagnostics: Once you arrive in Belgrade, we provide a comfortable transfer from the airport to our clinic or your accommodation. Day 1 includes basic tests and finalizing the treatment plan.
- Cell preparation: Before your arrival, our in-house laboratory team gets to work preparing donor stem cells for your child. We isolate and cultivate the MSCs and run them through our strict quality control tests (ensuring they are sterile, contaminant-free, and at the right potency and dose). After that, stem cells are ready for the treatment.
- Treatment sessions—the heart of care: Your child receives pain-free MSC infusions and exosome or macrophage therapy, often paired with supportive therapies like occupational or physiotherapy to nurture progress.
- Discharge & follow-up: Our support doesn’t end when your child’s treatment program finishes. Before you leave, we make sure everything is in order and, if needed, provide home-use exosomes to continue supporting your child’s progress. In the months that follow, we stay by your side with regular check-ins for at least three months, ensuring you feel guided, supported, and never alone on this journey.
How Successful Is Stem Cell Therapy for Autism?
One of the most common questions parents ask is, “What improvements can we expect to see, and how often does this actually work?”
While the success rate of stem cell therapy for autism varies by individual case, our data and experience show that the majority of patients will see at least a slight improvement in core autism symptoms.
Overall, approximately 80-90% of children after stem cell therapy for ASD demonstrate progress in at least one core area—social or behavioral skills. Improvements range from small but life-changing (e.g., fewer tantrums, better sleep) to major milestones like speaking first words or gaining new learning abilities.
For a deeper explanation of how we calculate success rates and what they really mean, read our dedicated article.
Read moreHow Long Does Stem Cell Therapy for Autism Last, and When to Expect Results?
Stem cell therapy doesn’t work like flipping a switch—it’s not an overnight change. We prepare parents for gradual improvement, which typically takes 3–6 months. Here’s what you can generally expect in terms of when improvements might appear and how long the benefits can last:
- First changes: Often seen within 1,5–3 months, sometimes even earlier (e.g., calmer mood, better sleep).
- Peak effect: Usually between 3–6 months, with progress continuing up to 12 months. Improvements may include better focus, calmer behavior, reduced irritability, improved sleep, easier eye contact and socialization, clearer speech, and fewer repetitive behaviors.
- Lasting benefits: Skills gained during this period are typically permanent, even after stem cells fade.
- Duration of treatment effects last 6–8 months, creating a window for faster learning.
Follow-up: Some families choose a booster treatment after 6–12 months to build on earlier progress.
Realistic outlook: When we talk about a “3–6 month peak” of improvement, it does not mean that after 6 months the child will regress. Any developmental milestones achieved, such as speech or self-care skills, are learned abilities that remain.
Stem cells open a window of opportunity for accelerated development, but it’s the ongoing teaching and therapy that translates that opportunity into permanent skills.
To maximize and maintain the progress after stem cell therapy, we advise families to keep up all traditional therapies:
- Behavioral therapy for autism to maintain and strengthen progress.
- Speech and language therapy for autism to improve communication skills.
What Are the Risks of Using Stem Cells to Treat Autism?
We understand that parents naturally worry about safety when considering any treatment for their child. The reassuring news is that MSC therapy for autism is known for its high safety profile.
In some cases, children may experience brief, mild reactions after stem cell therapy for ASD—like temporary fatigue, a low-grade fever, mild nausea, or a slight headache. These effects usually pass quickly and do not cause lasting discomfort. In our experience, no child has ever experienced long-term side effects, thanks to our strict safety protocols and the gentle, carefully monitored way we deliver each treatment.
If you’re interested in the technical side, you can discover how our lab produces high-quality stem cells while ensuring strict safety and purity standards in our detailed article.
Read moreOur Own Study: MSC Infusions in Children with ASD
We are not just a treatment center; we are also a research-oriented hospital. To continually improve our approach and contribute to the wider understanding of stem cell therapy for autism, we conducted an internal study on a group of our patients. In this study, we followed 30 children with ASD who received our MSC and exosome therapy protocol, and we tracked their progress over 9 months. Here are some key stem cell safety findings and outcomes from that study:
- No serious adverse events: Not a single child experienced a life-threatening or severe complication. There were zero cases of infusion-related infections, blood clots, organ damage, or hospitalization due to the therapy.
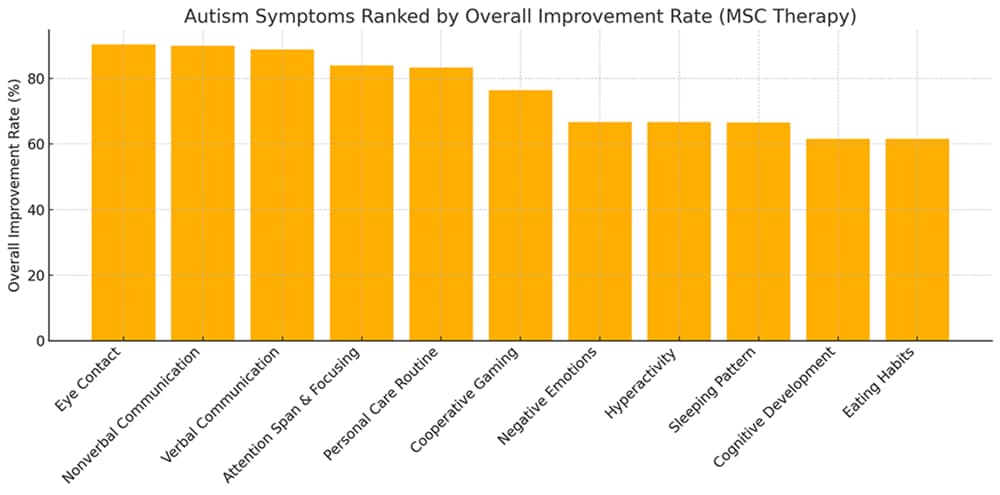
- Improvements noted: By 9 months, 93.4% had improvement in at least one symptom, and 83.3% improved in half or more of their symptoms.
To present a quick visual of the improvement rates from our study:
| Symptom/Benefit | Improvement Rate |
| Eye Contact | 90% |
| Nonverbal Communication | 90% |
| Verbal Communication | 89% |
| Attention & Focus | 84% |
| Personal Care Skills | 83% |
| Additional Benefits | Fewer seizures and infections in some children. |
Our Safety Protocols and Quality Measures
The safety of our stem cell therapy for autism isn’t accidental—it’s the result of carefully designed, rigorous protocols and strict quality measures that we follow at every step:
- In-house lab: 100% of our stem cells are prepared on-site in a sterile, controlled lab environment, minimizing contamination risks and ensuring fresh, potent cells.
- Quality testing: Each batch undergoes multiple safety checks (sterility, endotoxins, viability, genetic markers) before approval for use.
- Personalized dosing: Carefully calculated per kg of body weight to avoid overload or clumping, following evidence-based safety limits.
- Pre-treatment screening: Before a child is approved for therapy, we conduct thorough screening tests. Patients are screened for infections, organ function, and contraindications before therapy.
- Close monitoring during treatment: During the treatment, our medical team closely monitors each child’s vital signs and overall condition. A low patient-to-doctor ratio (just 3–4 children per doctor) allows for personalized attention not only during infusions but throughout the entire stay—so your child is always in expert hands.
If you’re interested in more details about our lab operations or want to understand how we implement safety protocols at a deeper level, we’ve put together an article that goes behind the scenes of our lab and safety practices.
Read moreReal Patient Stories
What matters most to us is seeing our patients make progress—step by step, change by change. To give you a real sense of what this journey can look like, here are a couple of patient stories told by their families in their own words:
“We were unsure. My husband and I came together the first time because we didn’t know what to expect. But we had nothing to lose.”
“One day after the second round, I saw our child put on his shoes by himself—for the first time ever. It may sound small, but for us, it’s huge. Swiss Medica has given us hope and real progress we never thought possible.”
— A parent from the UK
“We were losing him. He stopped talking, stopped calling us Mom and Dad. Nothing worked. After stem cell therapy at Swiss Medica, he opened his eyes and said his first word in years. Weeks later, he was talking to us, smiling, asking for hugs. It feels like we got our child back.”
— A mother of Yuval
How Much Does Stem Cell Therapy Cost for Autism?
We understand that cost is a significant consideration for any medical treatment, especially one that might not be available in your home country. Stem cell therapy is an advanced treatment requiring substantial medical expertise, cutting-edge lab facilities, and high-quality biological materials. However, Swiss Medica is committed to transparent, all-inclusive pricing, so you know exactly what you’re paying for, with no hidden fees.
At Swiss Medica, autism treatment packages typically range from €7,000 to €31,000*, depending on the program length and complexity of the case. This price includes not only the stem cell injections but also a full range of supporting services.
How much does stem cell therapy cost for autism in other countries? For context, in some countries a single round of stem cell therapy can cost $20,000 or more just for the cells, not including supportive therapies or accommodations.
*Prices are indicative, based on January 2025, and may vary with condition severity and cell quantity required.
What’s Included in Our Package
| Service | Included |
| Medical procedures (cells, lab work, infusions, supportive therapies) | ✓ |
| Pre- and post-treatment evaluations and lab tests | ✓ |
| Required medications and take-home exosome vials (when prescribed) | ✓ |
| Accommodation for child and one parent/caregiver | ✓ |
| Meals with special diet options | ✓ |
| Local transportation (airport and clinic transfers) | ✓ |
| 24/7 translator or personal assistant if needed | ✓ |
| Follow-up consultation | ✓ |
| Flights and personal expenses | ✗ |
Options for Payment & Travel Support
We don’t want logistical or financial hurdles to block a child from getting potential help. So we work hand-in-hand with you to handle details and find solutions that fit your situation:
- Payment plans and financial assistance
We are ready to discuss convenient payment options for our patients. While we do not provide direct financing, we can supply official cost letters and medical documentation to support applications for loans or fundraising in your home country. - Travel support
Our medical advisor can provide information about logistics such as flight options, airport transfers, and visas. Serbia allows visa-free entry for many countries; simply check the requirements for your country. - Accommodation adjustments
If you plan to arrive early or stay longer, we can provide you with a list of our partner hotels or assist you in arranging additional nights in our hospital.
Contact us
If you have concerns about cost or travel, just bring them up during the free consultation. We’re here to help address those and provide peace of mind along with the medical care.
Is Stem Cell Therapy for Autism Legal in the US and UK?
The legality and regulatory status of stem cell therapy for autism vary by country, and it’s a common point of confusion. In short, in the United States and United Kingdom, stem cell therapy for autism is not yet approved as a mainstream treatment—it’s considered experimental. What does it mean? Here’s a little more detail.
Current Regulation Overview
In the United States, the use of stem cells is strictly regulated by the U.S. Food and Drug Administration (FDA). Only a handful of stem cell treatments, primarily in hematology, have full FDA approval and must be administered in a hospital setting.
United Kingdom and EU: The European Medicines Agency (EMA) also requires rigorous trials.
A cautious regulatory stance often reflects the need for more long-term data—not a conclusion that the treatment is unsafe.
Stem cell therapy for autism is legal in the US/UK only in clinical trial contexts, not as a routine treatment. That’s why many families look internationally for treatment options—many countries have more permissive frameworks for innovative therapies.
Which Country Is Best for Stem Cell Therapy for Autism?
There isn’t a single best country for stem cell therapy for autism that fits everyone, but there are a few destinations that have become leading hubs—each with its pros and cons. This depends on factors like level of medical care, affordability, and comprehensive services for foreigners. Here are a few highlights:
- Serbia: Licensed clinics, strict protocols, experienced doctors, and affordable pricing. Easy access from Europe, visa-free for many nationalities, and extensive experience with autism cases.
- Mexico: Convenient for US families, with reputable clinics in Cancun, Tijuana, and Mexico City. Quality varies, so research is essential.
- Panama: Known for pioneering autism stem cell treatments. English-friendly but more expensive and less accessible for Europeans.
- India & Thailand: Lower-cost options with some reputable centers, but travel time and facility standards vary.


Ready to Get Started? Book Your Free Consultation
If you’re wondering about your child’s specific situation or asking yourself, “Does stem cell therapy work for autism in our case?” the best way to find out is to talk with one of our medical advisors. During our conversation, we’ll take the time to truly understand your child’s unique story, carefully review their case, and explain how treatment could be personalized to their needs. You’ll also have the chance to ask any questions on your mind.
Contact us
If you have concerns about cost or travel, just bring them up during the free consultation. We’re here to help address those and provide peace of mind along with the medical care.
You can also send us an email or message us via WhatsApp.
Frequently Asked Questions about Stem Cell Therapy for Autism
1. How many stem cell treatments will my child need?
Most children start with one treatment cycle (3–9 days on-site), followed by a 6-month evaluation period. A second or multiple cycles may be considered if progress plateaus.
2. My child is afraid of doctors. Will the procedure be painful or difficult for them?
We understand that many children—especially those with autism—feel nervous around doctors or medical procedures. That’s why we’ve designed our treatment process to be as gentle and child-friendly as possible.
The main stem cell procedure is done through a quick IV infusion, which feels like a brief needle prick. Other therapies, such as exosome inhalations and nasal sprays, are non-invasive and painless. Parents can stay by their child’s side the entire time, and our experienced staff uses calming, sensory-friendly techniques to create a low-stress environment.
3. What support will we receive during and after therapy?
Families receive 24/7 medical and emotional support during their stay. After treatment, we provide follow-up calls, ongoing guidance, and access to our team for questions or advice, ensuring long-term support.
4. Why are some doctors skeptical when I ask, “Can autism be treated with stem cells?”
Stem cell therapy is relatively new and not yet part of standard guidelines in many countries. Some doctors lack updated knowledge or recall past unregulated practices. Skepticism usually reflects caution, not proven ineffectiveness. We share data and results with doctors to help bridge this gap.
5. Can stem cells cure autism?
“Can autism go away?” is a question that many patients ask. There’s no known cure for autism. Therapy aims to reduce symptoms and improve quality of life.
6. Can we continue other therapies alongside stem cell treatment?
Yes, and it’s encouraged. Stem cell therapy often enhances responsiveness to ABA, speech, OT, and other interventions. Most medications and supplements can continue without conflict.
7. Is stem cell therapy for autism legal and safe?
At our clinic, stem cell therapy for autism is the main focus. All treatments follow strict safety protocols. MSCs have an excellent safety record with minimal side effects like mild fever or nausea. We thoroughly screen patients to ensure suitability.
List of References:
Akat, A., Karaöz, E. Cell therapies for autism spectrum disorder: a systematic review of clinical applications. Middle East Curr Psychiatry 30, 94 (2023). https://doi.org/10.1186/s43045-023-00363-9
Eve Madeline , Gandawijaya Josan , Yang Liming , Oguro-Ando Asami. Neuronal Cell Adhesion Molecules May Mediate Neuroinflammation in Autism Spectrum Disorder. Frontiers in Psychiatry. Volume 13 – 2022. doi.og/10.3389/fpsyt.2022.842755
Liang Z, Zhang G, Gan G, Naren D, Liu X, Liu H, Mo J, Lu S, Nie D, Ma L. Preclinical Short-term and Long-term Safety of Human Bone Marrow Mesenchymal Stem Cells. Cell Transplant. 2023 Jan-Dec;32:9636897231213271. doi.org/10.1177/09636897231213271
Gesundheit B, Hochbaum L, Fetyukhina A, Vorobyev V, Vorobyev N. Mesenchymal Stromal Cell Treatment Alleviates Autism Spectrum Disorder Symptoms: A Case Report. Cureus. 2025 May 4;17(5):e83440. doi.org/10.7759/cureus.83440
MD, Pediatrician, Regenerative Medicine Specialist