Stroke can abruptly damage areas of the brain responsible for movement, speech, memory, and cognition. Many survivors struggle with lasting impairments despite months of rehabilitation.
Stem cell therapy for stroke patients is emerging as a supportive regenerative approach: it helps reduce inflammation, improves the brain’s microenvironment, and strengthens the natural recovery processes that continue long after the acute phase. At Swiss Medica, we use stem cells and complementary biological products to support post-stroke rehabilitation and help patients regain mobility, clarity of speech, and independence.
Understanding Stroke and Its Aftereffects
A stroke occurs when blood flow to part of the brain is interrupted, depriving neurons of oxygen and nutrients.

What happens in the brain after a stroke
Some cells die immediately, while surrounding areas become inflamed and lose normal function. These “injured but not dead” regions often retain partial ability to recover if the microenvironment is stabilized and inflammation is reduced.
Common complications—mobility, speech, and cognition
Post-stroke impairments vary depending on the location and size of the injury:
- limb weakness, imbalance, poor coordination and falls
- difficulties with speech (aphasia), articulation, or comprehension
- memory lapses, slower information processing, reduced attention and planning
- emotional instability, anxiety, depression or fatigue
These symptoms often persist even with intensive rehabilitation, particularly in people with chronic stroke.
Why standard rehabilitation alone may not be enough
Conventional physiotherapy and speech therapy help retrain existing neural pathways, but they cannot directly improve the biological condition of injured tissues. When inflammation remains high and metabolic processes are disrupted, progress slows or plateaus. This is where regenerative stem cell–based treatment after stroke can be introduced—not instead of rehabilitation, but alongside it—to stabilize the neuronal environment and support functional recovery.
How Does Stem Cell Therapy Work for Stroke Patients?
Stem cells can activate the body’s natural repair processes by affecting surrounding cells and tissues with secreted molecules.
Brain tissue damaged by ischemia can retain its ability to restore structure and function, which is indirectly aided by stem cells.
How stem cells support brain repair and neuron regeneration
Stem cells do not simply replace dead neurons. Instead, they enhance the brain’s internal repair capacity:
- release growth factors that support surviving neurons
- stimulate local stem cells in the brain
- promote angiogenesis, improving blood supply to injured areas
- help restore connections between neural networks
In other words, when we use stem cell therapy to repair stroke damage, we support the body’s own healing mechanisms rather than “growing a new brain”.
Reducing inflammation and improving circulation
After a stroke, inflammation can become one of the major obstacles to recovery. Mesenchymal stromal cells (MSCs)—among the most effective stem cells for stroke recovery—help regulate the immune response and lower inflammatory cytokines. Better microcirculation and reduced swelling provide remaining neurons a more favorable environment to function and reorganize.
Enhancing the brain’s natural healing and plasticity
Plasticity is the brain’s ability to form new pathways. By stabilizing tissue and improving oxygenation, stem cells for stroke recovery may enhance neuroplastic potential, making physiotherapy and cognitive training more effective.
This is why stem cells for stroke are being explored as an adjunct method—not a cure, but a biological “boost” for damaged neural circuits.
Types of Stem Cells Used in Stroke Recovery
At Swiss Medica, we use only adult multipotent mesenchymal stromal cells (MSCs) and do not work with embryonic or induced pluripotent stem cells.
Mesenchymal stem cells
MSCs are the most widely studied and used cell type in stem cell treatments for stroke. These cells have several important properties:
- help regulate inflammation
- support tissue repair
- create favorable conditions for functional improvement
- are generally well tolerated in clinical trials.
Placental MSCs, exosomes and macrophages for enhanced neuroprotection
Our approach may also include advanced biological products designed to enhance stem cell treatment for stroke patients:
- exosomes—cell-derived vesicles carrying proteins and RNA, used intravenously and as nasal sprays to support neuroprotection
- secretome—active biological molecules that participate in cell-to-cell communication and healing processes
- macrophages (M2 phenotype)—immune cells helping clear debris and support tissue stability
- neural stem cells in specific protocols—to enhance regenerative signals in neurological conditions
These methods are based on the actual clinical practice at Swiss Medica and help broaden the therapeutic effect of stem cell injections for stroke patients.
Scientists worldwide are also studying neural progenitors and iPSC-based cells, but they remain experimental and are not part of Swiss Medica therapies.
Can Stem Cells Repair the Brain After Stroke?
Stem cells cannot fully regenerate dead brain tissue. Instead, stem cells for stroke survivors help restore functional communication between neurons that are still alive but weakened. This “functional recovery” often translates into:
- improved arm and leg mobility
- clearer speech and better articulation
- enhanced focus, memory, and mood
- more stable energy levels and better tolerance of exercise
Current evidence suggests it may support rehabilitation, especially when combined with targeted therapies, but results vary between individuals.
Safety of Stem Cell Therapy for Stroke Patients
To ensure stem cell therapy is delivered as safely as possible, Swiss Medica uses a structured safety protocol that includes:
- only adult donor MSCs from placenta and umbilical cord are used
- all products undergo strict sterility, viability, and identity testing
- procedures follow EU-aligned regulations and are performed by trained medical staff
- doses are calculated individually; we never exceed a maximum safe dose
- side effects, if present, are usually mild: short fever, fatigue, or local redness at the injection site
These factors make stem cell therapy a generally well-tolerated option for eligible post-stroke patients, although no medical procedure is entirely risk-free.
Clinical Evidence and Research on Stem Cell–Based Treatment After Stroke
Most patients who receive stem cell–based treatment after stroke and continue rehabilitation notice some degree of change over the following months, but the level of improvement is individual.
| Motor function and mobility | Many trials report modest but measurable gains in motor scores and walking ability when stem cells are added to standard rehabilitation. |
| Daily activities and independence | Research shows better performance on scales that measure everyday activities (such as dressing, bathing, transfers) in a portion of treated patients. |
| Speech, cognition, and energy | Clinical data are more limited, but some trials and case reports describe improvements in attention, processing speed, and global functional scales; patients often report better mental clarity, mood, and fatigue levels. |
Researchers are still refining protocols and analyzing the success rate of stem cell therapy for stroke, but many trials confirm a favorable safety profile and measurable functional gains in at least part of the treated patients.
Benefits of Stem Cell Therapy After Stroke
Based on scientific literature and Swiss Medica outcomes, stem cell therapy for stroke patients and rehabilitation may offer the following potential benefits:
- Some improvement in strength, control, balance, or walking (with or without aids).
- Greater independence in basic self-care and household tasks, with less hands-on help from family or caregivers.
- Clearer speech, better ability to find words, improved focus during conversation, and more energy for rehabilitation and daily life. Changes are usually gradual and depend strongly on the extent and location of brain damage.
Real-world patient improvements
Five years after stroke—Nicoleta’s father from Romania
After a stroke, Nicoleta’s father was left with right-side paresis, walked with a stick, and still depended on his family for many daily tasks like cooking and cleaning. Over time, his speech improved to about 70–80%, but progress gradually plateaued despite multiple rounds of rehabilitation. Some doctors eventually told the family that there was “no chance” for further recovery, which pushed them to start seeking options abroad.
Nicoleta and her father found Swiss Medica through online reviews and video testimonials from other stroke patients. They decided to come for an eight-day treatment course combining cell-based therapy and rehabilitation.
Nicoleta describes the stay as being “above expectations”—accommodation similar to a five-star hotel and staff who were always nearby, making them feel safe and supported throughout the procedures.
“Even when doctors at home said there was no chance, we decided not to give up. We saw other patients’ stories, came here, and felt truly cared for every day.”
Eight years after stroke—Kyle from Australia
A stroke left Kyle with weakness in his left arm and hand and sensory problems in his arm and leg. He lost his truck-driving license and, with it, the job he had done for more than 20 years. In his words, “In Australia they had nothing” more to offer for his condition, and he did not receive any advanced options beyond basic care.
Kyle decided to travel to Serbia for a two-week course of stem cell–based treatment and intensive rehabilitation at Swiss Medica. He says he wasn’t worried about coming to another country; he “just wanted to try it and hope for the best.” During his stay, he highlights how “the doctors are unreal,” how the staff explain everything clearly even if English is difficult, and how the food and care made him feel completely looked after.
Kyle also noticed early changes: a return of sensation in his arm and leg within the first days after the procedure, while understanding that the main results are expected over the next several months.
“If you’re thinking about it and you’re not sure, you really get one chance to try. I’d recommend doing it straight away rather than waiting and wondering.”
Individual results vary and depend on stroke severity, time since stroke, overall health, and engagement in rehabilitation. These two stories are just a small part of our experience—there are over 600 patient testimonials for different conditions on our official YouTube channel.
Combining Stem Cells with Rehabilitation
While stem cells improve the biological environment, rehabilitation drives neuroplasticity; Together, they create a synergistic effect:
- physiotherapy and kinesiotherapy strengthen neural pathways
- speech therapy improves communication circuits
- occupational therapy re-establishes daily skills
- hardware physiotherapy, super inductive system, and other modalities stimulate muscles and nerves
- MSCs help reduce inflammation and stabilize neurons
For this reason, stem cells for stroke symptoms are not used in isolation. They are integrated into a broader program that also addresses muscle strength, posture, coordination, cognition, and emotional health.
Contact us
Interested in treatment and want to know if stem cells for stroke could be suitable in your case?
Leave your contact information, and our medical team will reach out to schedule a free consultation.

Medical Advisor, Swiss Medica doctor
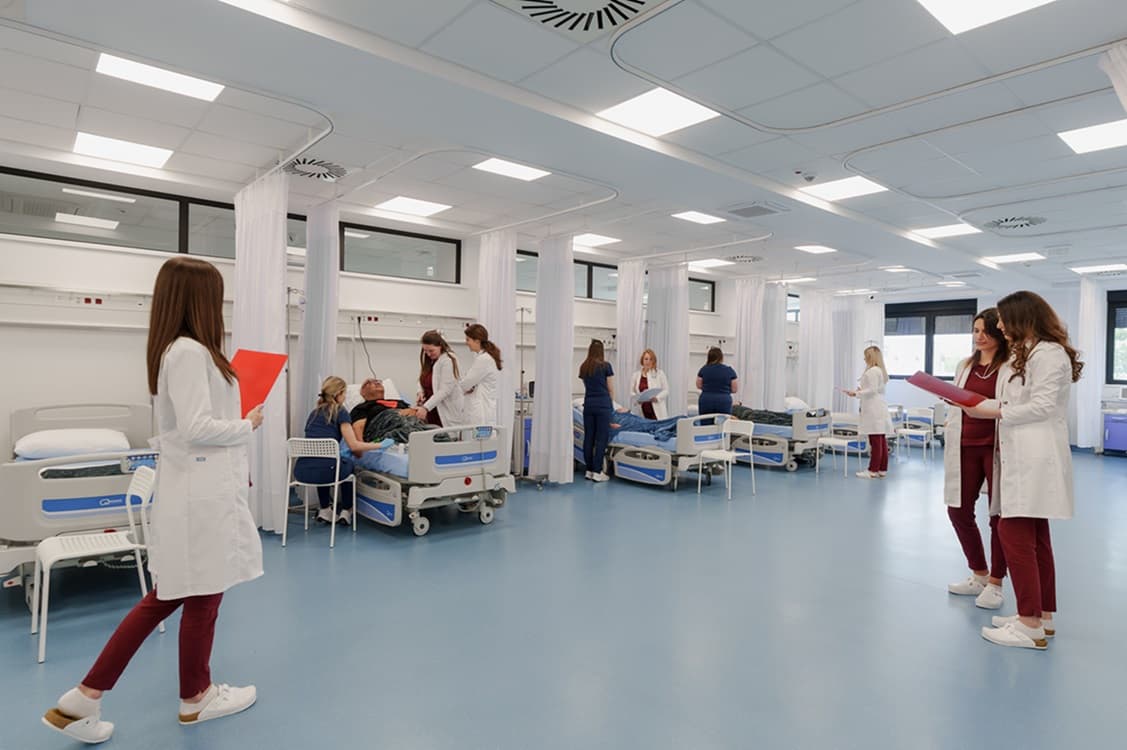
Swiss Medica Hospital in Belgrade: Modern Facilities for Stroke Patients
Swiss Medica’s hospital in Belgrade was designed specifically with neurological and post-stroke patients in mind. The campus includes several interconnected buildings with extensive medical and rehabilitation space, an in-house laboratory, and comfortable patient rooms that feel more like a boutique hotel than a traditional hospital.
For people with limited mobility after stroke, accessibility is critical. The hospital is fully adapted for wheelchair users and patients who need support:
- step-free entrances, ramps, and wide corridors
- elevators connecting all key areas of the clinic
- barrier-free bathrooms and showers, equipped with grab bars and seats
- height-adjustable beds and call systems for quick assistance
This setup makes daily activities—getting out of bed, going to the bathroom, taking a shower—safer and less stressful for patients and their families.
The Swiss Medica Stem Cell Treatment Process
Step 1—Diagnostic assessment and neurological evaluation
Doctors review medical records, MRI or CT scans (if available), comorbidities, medications, and stroke history to determine eligibility. This is also when many patients ask whether stem cell treatment for stroke is suitable for their particular situation. The answer depends on diagnosis, overall health, and realistic expectations.
Step 2—Cell preparation and administration
Stem cells are cultivated in our in-house lab under strict control. Depending on the protocol, they are administered:
- intravenously
- intrathecally, if recommended
- in combination with exosomes or other derivatives
This structured approach is sometimes described as a stem cell transplant for stroke, although we do not replace the entire blood or immune system as in classic bone marrow transplantation.
Step 3—Integration with physiotherapy and neuro-rehabilitation
During the in-clinic stay (usually 5–9 days), patients undergo an intensive rehabilitation program coordinated with cell therapy:
- kinesiotherapy and gait training
- balance and coordination exercises
- speech and cognitive therapy
- intracellular metabolism recovery therapy (IMR)
- super inductive system, shock/spark wave and other device-based therapies
By combining biological support with functional training, this regenerative approach becomes part of a larger, goal-oriented recovery plan. In some cases, additional booster courses of stem cell treatment for stroke patients can be considered.
Step 4—Post-therapy monitoring and progress tracking
For several weeks after discharge, many patients receive exosomes at home to support the effect of MSCs. Doctors follow up regularly, adjusting recommendations based on progress. This long-term approach helps optimize real-life outcomes of such regenerative therapy, beyond short-term clinical scales.
Aftercare and Follow-Up Program
After the main course of treatment, Swiss Medica provides:
- exosome nasal spray for home use
- regular doctor assessments
- detailed home-based rehabilitation recommendations
- optional free check-up to evaluate progress
Why Choose Swiss Medica for Stroke Recovery
Many patients choose Swiss Medica not only for the therapy itself but also for the environment in which it is delivered.
- Experience in regenerative medicine since 2011, with a special focus on neurological conditions and post-stroke recovery.
- Multidisciplinary team: neurologists, rehabilitation specialists, nurses, and cell therapy experts collaborate on each case instead of working in isolation.
- In-house A-grade laboratory and cryobank: all cell products are processed, tested, and stored on-site, which permits the medical team full control over quality and timing. There is no need to rely on external labs or long-distance transport.
- Focused attention: the doctor–patient ratio is intentionally low—around 3–4 patients per doctor.
- 50+ methods of rehabilitation and supportive care.
All-inclusive stay: treatment price typically covers accommodation, meals (including special diets), care for one accompanying person, transfers to and from the airport and interpreter services. This is especially important for international patients who may not be familiar with the local healthcare system.
The hospital setting is designed to facilitate treatment and minimize physical and emotional stress.
Patients can concentrate on rehabilitation while the team takes care of logistics, safety, and comfort—factors that often have a direct impact on how well people engage in therapy and maintain progress after a stroke.
Stem Cell Therapy for Stroke: Costs and What Affects Them
When individuals and their families are navigating a serious health diagnosis, the search for effective treatment often leads them to look beyond their home countries. In this journey, the research path is often marked by a few critical questions that quickly move from the general to the deeply personal. People want to know the best country to get stem cell therapy; they also need to understand how much stem cell therapy costs.
At Swiss Medica the prices of stem cell therapy range from €7,000 to €31,000*. The final cost of stem cell therapy for stroke depends on:
- the number of procedures and days in the clinic
- the quantity of cells required
- the patient’s age, weight, and overall health
- additional therapies (physiotherapy, device-based treatments, IMR)
- need for follow-up visits or booster courses
Serbia often offers more accessible pricing compared to the US or Western Europe while maintaining high safety and quality standards. Exact figures can only be given after an individual medical evaluation.
*Prices are indicative, based on January 2025, and may vary with condition severity and cell quantity required.
Getting Started with Consultation
If you are wondering whether stem cell therapy could help in your specific case, the first step is a detailed consultation:
- Book a free online consultation with a regenerative medicine specialist.
- Send your medical records and imaging results for preliminary review.
- Discuss whether a regenerative stem cell–based approach after stroke is appropriate in your case and what realistic goals may look like.
- Receive a personalized treatment and rehabilitation plan, including approximate stem cell therapy for stroke cost and logistics.
Contact us
Ready to contact us for a no-obligation discussion to help you decide whether traveling for a stem cell–based stroke treatment is a reasonable option? Just fill out the form below.

Medical Advisor, Swiss Medica doctor
Frequently Asked Questions
1. Can stem cells repair the brain after a stroke?
No, stem cell treatments for stroke do not regenerate dead tissue, but they can improve the condition of surviving neurons and support functional recovery.
2. Is stem cell therapy good for stroke patients of all ages?
Age is only one factor. The consulting doctor will consider overall health, stroke type, time since the event, and current level of function before recommending any advanced stem cell treatment for stroke.
3. What is the success rate of stem cell therapy for stroke?
There is no universal number. Clinical trials show encouraging functional gains in many participants, but outcomes vary. The question “how successful is stem cell transplant or any other cell-based stroke procedure?” will always have a personalized answer based on your diagnosis and goals.
4. Can stem cells help stroke ‘victims’ more than standard treatment?
No. Stem cell therapy for stroke ‘victims’ does not replace standard treatment, rehabilitation, or risk factor control. It is an additional option that may enhance recovery and quality of life when used in a structured program.
List of References:
Chang, W., Ma, X., Zhang, H. et al. The efficacy and safety of stem cell therapy for ischemic stroke: a systematic review and network meta-analysis study. BMC Neurol 25, 235 (2025). https://doi.org/10.1186/s12883-025-04246-w
Hassanein Maged Elsayed , Fayad Jaber , Shabana Jilan Ali , AlEdani Esraa M. , Hefnawy Mahmoud Tarek , Ghaith Hazem S. , Negida Ahmed. Efficacy and safety of intravenous mesenchymal stem cells for ischemic stroke patients, a systematic review and meta-analysis, 2024. https://www.frontiersin.org/journals/stroke/articles/10.3389/fstro.2023.1339331
Choudhery, M.S.; Arif, T.; Mahmood, R.; Harris, D.T. Therapeutic Potential of Mesenchymal Stem Cells in Stroke Treatment. Biomolecules 2025, 15, 558. https://doi.org/10.3390/biom15040558
Thomas L, Rana S, Duong A, Kirkham AM, Shorr R, Maganti H, Seftel M, Allan DS. The emerging promise of induced pluripotent stem cells in clinical studies: a systematic scoping review of the literature and registered clinical trials. Cytotherapy. 2025 Sep 26:S1465-3249(25)00846-1. doi: 10.1016/j.jcyt.2025.09.008. Epub ahead of print. PMID: 41160002.
MD, Pediatrician, Regenerative Medicine Specialist