Motor neuron disease (MND) is a group of progressive neurodegenerative conditions that selectively affect the upper motor neurons (UMN) and lower motor neurons (LMN), leading to muscle weakness, atrophy, and impaired voluntary movement.
As motor neurons stop working, muscles weaken, and everyday functions—walking, speaking, swallowing, and breathing—can become harder to maintain.
Standard care focuses on slowing progression where possible and protecting quality of life with coordinated support. However, researchers are also exploring stem cell treatment for motor neuron disease as a supportive option—often discussed as a potential new treatment for MND focused on neuroprotection and inflammation control.
What Is Motor Neuron Disease?
MND is an umbrella term for disorders that gradually damage motor neurons in the brain (UMN) and spinal cord (LMN). Because these neurons control muscle activity, their loss disrupts communication between the nervous system and muscles.
How motor neuron disease affects the nervous system
Upper motor neurons help regulate movement from the brain, while lower motor neurons carry signals from the spinal cord and brainstem to muscles. When these pathways degenerate, muscles may become weak, stiff, or uncoordinated, and muscle bulk can decrease over time.
Types of MND (ALS, PLS, PMA, SMA)
Similar conditions are classified into groups based on the extent, speed, and cause of damage to the nervous system. Typically mentioned subtypes include:
- ALS (amyotrophic lateral sclerosis): affects both upper and lower motor neurons and often progresses faster than other adult MND subtypes.
- PLS (primary lateral sclerosis): involvement of upper motor neurons and often a slower course.
- PMA (progressive muscular atrophy): involvement of lower motor neurons, with variable progression.
Symptoms and Disease Progression
The progressive loss of independence as symptoms spread throughout the body is a common way to describe the journey of MND.
Early-stage symptoms
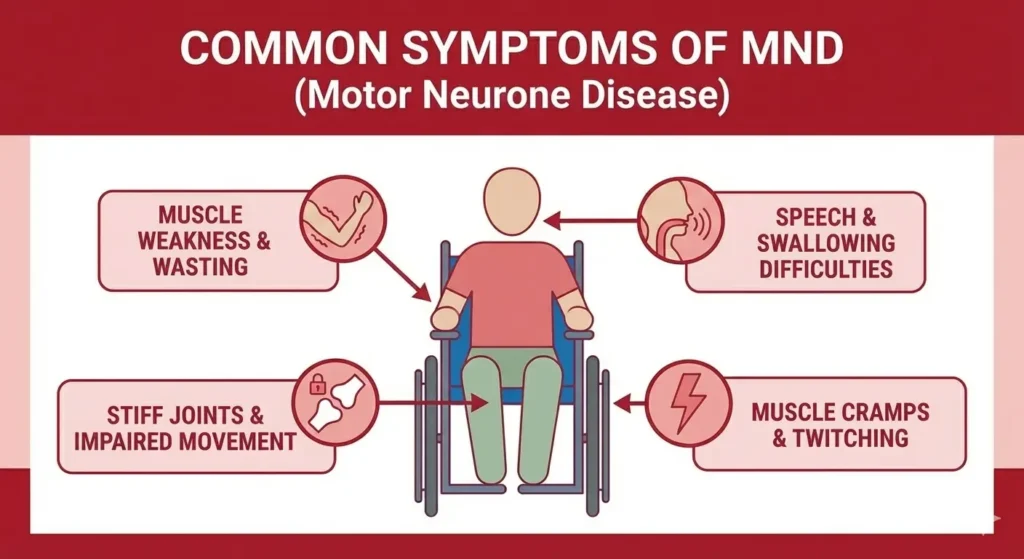
Early symptoms can be subtle and may start in one region:
- weakness in one hand, arm, or leg (grip, fine motor tasks, foot drop)
- balance changes or reduced coordination
- cramps, stiffness, or muscle twitching
- early speech or swallowing strain
Advanced-stage complications
As MND progresses, symptoms may involve several domains:
- reduced mobility and higher falls risk, with increasing need for assistive devices
- weakened cough reflex and reduced breathing reserve, especially during sleep
- speech and swallowing difficulties, aspiration risk, weight loss
Current Treatment Approaches and Their Limitations
Standard medical care for MND includes medications and supportive measures that are primarily palliative in nature, aiming to manage symptoms and slow disease progression.
Disease-modifying and symptomatic medications
The primary drug, riluzole, may extend life by modulating glutamate levels, but it does not stop neurodegeneration. Other medications manage secondary symptoms like muscle cramps and excessive salivation.
Supportive, rehabilitative, and palliative care
Rehabilitation, nutrition, respiratory monitoring, and palliative support help protect function and reduce complications. Physiotherapy and occupational therapy are vital for maintaining independence.
Traditional methods are essential for managing MND, but they have limited ability to address the underlying biological stress and degeneration affecting the nervous system. This has led to growing interest in alternative treatments for motor neuron disease, such as regenerative stem cell therapy.
Why Regenerative Therapies Are Being Studied for MND
Regenerative medicine is being studied because MND is not driven by a single mechanism, and supportive biological signals may matter alongside symptom control. In that sense, some researchers describe these approaches as a new treatment for MND within the field of supportive care, rather than a stand-alone solution.
Motor neuron loss and lack of natural regeneration
Unlike skin or bone, the central nervous system has a very limited ability to repair itself. Once a motor neuron dies, it is not naturally replaced. Regenerative medicine seeks to change the “microenvironment” of the spinal cord to make it more hospitable for surviving cells.
The role of inflammation and immune dysregulation
Many neurodegenerative conditions involve chronic inflammation and immune signaling changes. Research interest in this area has contributed to studying cell-based approaches that may modulate inflammatory pathways.
Potential for neuroprotective support
The core scientific idea behind many regenerative medicine protocols is “support the environment around neurons”. This is why stem cell therapy for MND is framed as a supportive strategy designed to actively stimulate the neural microenvironment. By regulating inflammation and delivering protective signals, the therapy aims to enhance neuronal survival and create the biological conditions necessary to sustain and amplify rehabilitation efforts.
Can Stem Cells Cure Motor Neuron Disease?
Families often ask: can stem cells cure motor neurone disease? Unfortunately, the evidence does not support a cure claim.
What “cure” means in neurodegenerative conditions
A cure would mean stopping the disease process and reliably restoring lost motor neuron function across patients. That standard has not been met.
Realistic therapeutic goals for MND patients
When stem cell therapy for MND is discussed responsibly, goals are supportive and include:
- supporting function and day-to-day stability
- potentially slowing decline in selected cases
- reducing inflammation-related symptom burden
- improving rehabilitation response and quality of life
In other words, stem cell therapy is not curative. While it is considered an alternative treatment for motor neurone disease, it is most effective when integrated with conventional methods. Rather than choosing one over the other, the goal is to combine these strategies to enhance overall results while the patient continues their standard neurological care.
Stem Cells for Motor Neuron Disease: How They Work
Stem cells are specialized cells found throughout the body that have the unique ability to differentiate into different types of tissue when stimulated. The cells used in therapy are valuable because they release molecules that create a natural environment conducive to regeneration.
Types of Stem Cells Used in MND Therapy
At Swiss Medica, we use only adult multipotent mesenchymal stem cells (MSCs) and do not use embryonic cells (ESCs) or induced pluripotent stem cells (iPSCs).
Donor-derived MSCs are typically sourced from ethically obtained umbilical cord or placental tissue, with consent and screening.
Autologous MSCs may be used in selected cases. In this situation, the cells are sourced from the patient’s own body, either from bone marrow or adipose tissue.
Autologous NSCs may be used in specific clinical situations depending on the patient’s profile.

Other biomedical products, such as exosomes (signaling vesicles), are also used to enhance these effects.
Stem Cells for Motor Neuron Disease: Mechanisms of Action
In people with MND, MSCs may support the nervous system through complementary mechanisms:
Neuroprotection
- MSCs and NSCs can release growth factors such as BDNF, GDNF, and VEGF that help motor neurons survive and function longer. These factors can reduce oxidative stress and improve mitochondrial function, both of which are disturbed in MND.
Immunomodulation (immune calming)
- MSCs can dampen overactive immune responses and reduce harmful inflammatory cytokines (for example, TNF‑α, IL‑6) that contribute to motor neuron injury. This may help create a more balanced environment in the spinal cord for existing neurons.
Anti-apoptotic and cell-survival signaling
- Stem cells can secrete molecules that interfere with programmed cell death pathways, potentially slowing motor neuron loss.
How Regenerative Treatment for MND Is Performed
At Swiss Medica, stem cell therapy for MND is delivered within a structured hospital-based program, integrating regenerative support with monitoring and rehabilitation.
Cell sourcing, preparation, and quality control
When donor-derived MSCs are used, the tissue source is typically umbilical cord or placental tissue obtained with consent. Cells are expanded in a sterile in-house, GMP-certified laboratory. Before use, quality control steps include sterility testing and screening for contamination (bacterial, fungal, and viral), as well as checks for identity and viability.
Routes of administration and monitoring
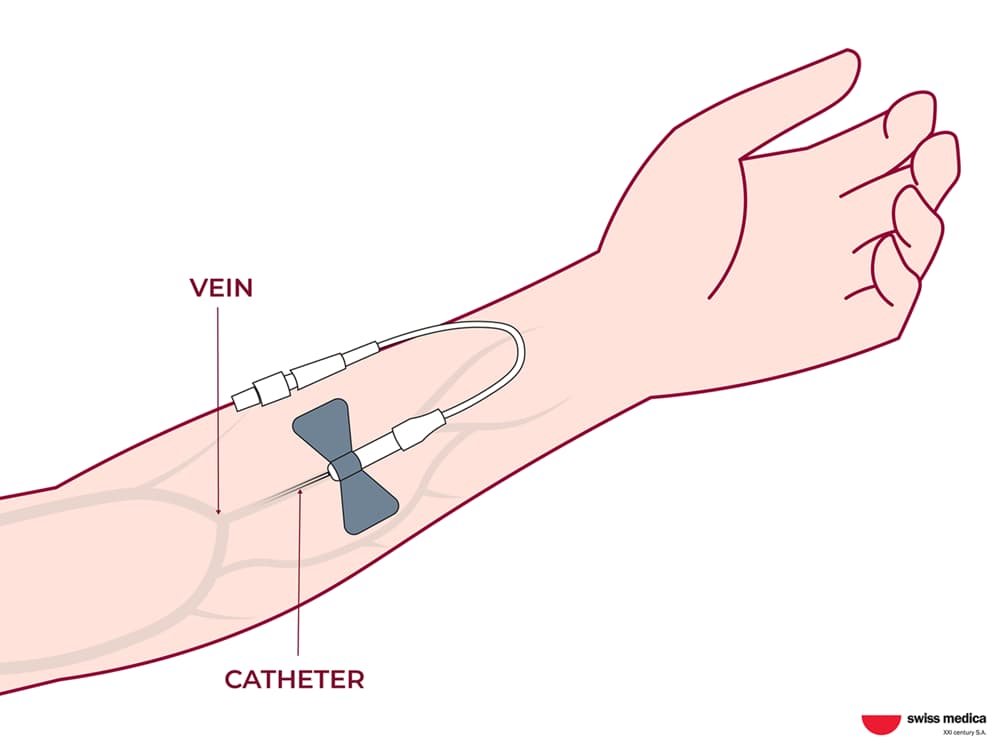
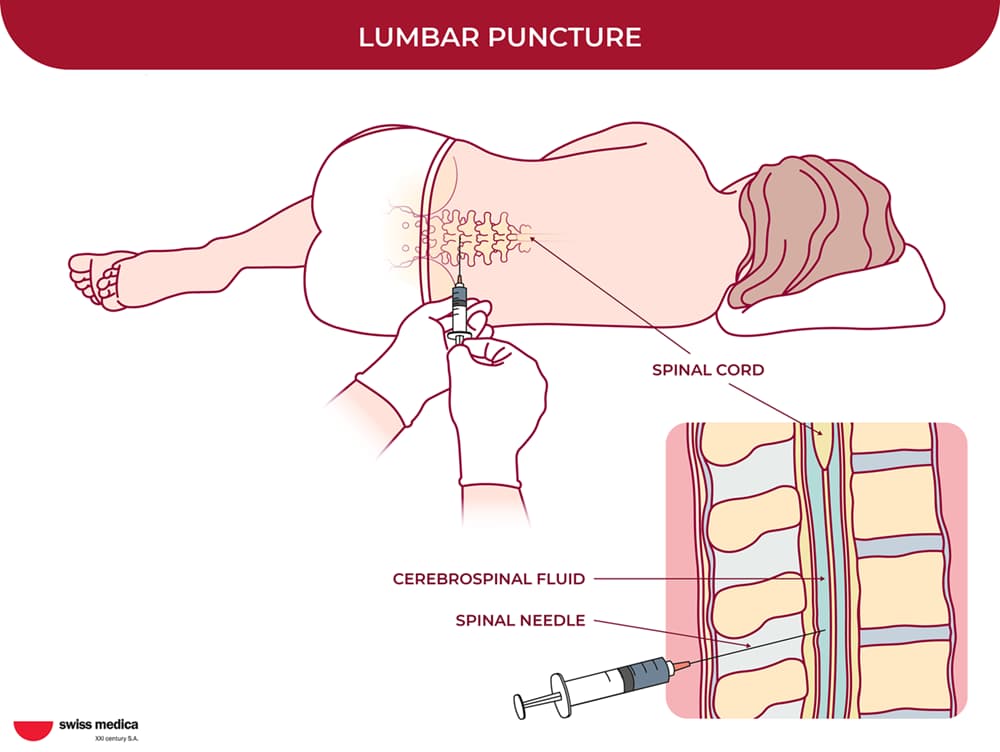
Administration routes are selected by the physician based on clinical rationale and safety considerations. Depending on the case, MSCs can be delivered in two different ways:
- intravenously (IV) for more systemic support
- intrathecally (into the cerebrospinal fluid) for introducing cells directly to the affected area.
Vital signs are monitored by a multidisciplinary team to ensure safety and comfort during administration, and clinical observation continues afterward.
Safety, Risks, and Clinical Evidence on Stem Cell Treatment for Motor Neuron Disease
Adult MSCs have a well-documented safety record, and the most common adverse effects are mild, such as temporary local swelling or a low-grade fever following injection.
Several phase I/II studies of stem cell therapy for ALS also report that the procedures are feasible and generally well tolerated, with similar overall adverse event rates between the MSC and control groups and no clear excess of serious treatment-related complications.
Risks can become significantly higher when stem cell treatments are delivered in uncertified or poorly regulated clinics that do not follow established manufacturing, sterility, and monitoring standards.
Safety protocols at Swiss Medica
At Swiss Medica, the safety of our patients is our top priority. We have strict safety protocols in place for neurological patients, which typically emphasize:
- detailed pre-admission medical review and risk stratification
- cells infection screening and contraindication checks
- supportive therapies adjusted to fatigue level, mobility, and respiratory status
Learn more about safety standards at Swiss Medica, including in-house laboratory controls and cell production processes.
Discover nowPotential Benefits Patients May Experience After Stem Cell Therapy for Motor Neurone Disease
When assessing stem cell therapy for motor neuron disease, it is crucial to analyze its comprehensive effects on the patient. Initial changes may become apparent within a few weeks, with optimal therapeutic effects generally observed between 3 and 6 months following treatment.
Possible slowing of functional decline
Some patients report that endurance for walking or daily tasks declines more slowly for a period of time.
Symptom support and quality-of-life improvements
Increased endurance during rehabilitation, more stable mobility during transfers, and less weariness are sometimes reported by patients. Enhancements in limb strength, speech clarity, or ease of breathing, can have a big impact on a patient’s independence and mental health.
What stem cell therapy cannot change
Although some studies describe that mesenchymal stem cells can transform into neurons and replace damaged tissue, these conclusions are exaggerated. Stem cells cannot replace an entire organ or tissue, but due to their ability to activate the body’s reserves for regeneration, they can improve the prognosis of chronic diseases and promote neuron recovery.
Importance of ongoing standard care
Standard neurological management, respiratory planning, nutrition strategies, assistive devices, and rehabilitation remain essential even when stem cell therapy for MND is added.
Marie From the UK—Experience with Stem Cells
Marie came to Swiss Medica from the UK after searching for a stem cell treatment center for motor neuron disease. She felt that, at home, the medical system offered little more than a “waiting game.” After her diagnosis, she struggled with frequent falls, weakened breathing, and slurred speech. However, the changes were visible after the regenerative program at Swiss Medica. Marie’s family noted the following improvements:
“Breathing, swallowing, and speech… already improved a little bit. Walking is better, and she can move her toes now, which she couldn’t before.”
Although individual results may vary, real-world evidence often speaks louder than statistics. Consider the experience of Marie and over 500 video testimonials relating to various conditions on our official YouTube channel.
Who May Be a Suitable Candidate for Stem Cell Treatment for Motor Neuron Disease
A reputable evaluation focuses on safety first, then realistic goals. When selecting a stem cell treatment center motor neuron disease program, careful screening protects patient safety and supports appropriate expectations.
A patient may be considered based on:
- presence of a diagnosis
- disease stage and rate of progression
- respiratory status (breathing tests, ventilation needs)
- swallowing safety and nutrition stability
- overall medical stability (cardiovascular status, infection risk)
A patient may not be eligible if any of the following conditions are present:
- advanced bulbar impairments, including severe swallowing difficulties and high risk of aspiration.
- advanced respiratory failure requiring complex ventilatory support that makes travel unsafe
- active infection
- active malignancy or recent cancer history
- medical instability that increases procedural risk
Get a free online consultation
Each case is unique. You can book a free online consultation with our regenerative medicine doctor to find out if your condition is suitable for treatment.

Medical Advisor, Swiss Medica doctor
The Swiss Medica Approach to Stem Cell Treatment for Motor Neuron Disease
Swiss Medica is a stem cell clinic where regenerative medicine is integrated with rehabilitation and supportive care. For patients looking at different treatment options, the key question is whether the stem cell treatment center provides complete medical oversight, safe preparation of biomedical products, and a range of support services all in one place.
Multidisciplinary neurological care
Our team coordinates neurological assessment with regenerative and symptom-focused medical care so functional goals remain practical and measurable.
Personalized regenerative protocols
Every protocol is developed based on the patient’s symptoms, goals, and general health.
Supportive rehabilitation and monitoring
Swiss Medica enhances the overall outcomes by combining supportive therapies with stem cell therapy. In the course of the MND program, patients might get:
- PRP therapy: Concentrated platelets taken from a patient’s own blood can promote healing and reduce inflammation.
- Kinesiotherapy and physiotherapy: A personalized movement-based rehabilitation designed to improve physical potential and address functional limitations. Under expert guidance, patients perform tailored programs involving voluntary dynamic movements and static muscle contractions. These sessions aim to increase strength, endurance, and mobility for daily activities.
- Advanced device-based therapies: Including electro/electromagnetic stimulation (SIS), targeted ultrasound treatment, and shockwave therapy to stimulate nerve and muscle response.
- Exosome therapy: Tiny vesicles derived from stem cells that improve tissue repair and transmit regenerative signals.
Prior to discharge, the team reevaluates your condition, reviews the post-care instructions, and confirms your understanding of how to proceed with your rehabilitation at home.
Follow-up
Upon completion of the inpatient program, you will be provided with a supply of exosomes for home use. This supply is intended to support your recovery for approximately two months and marks the conclusion of the treatment materials included in the initial program.
Following your return home, the clinic remains in contact with you by arranging follow-up appointments at 3 and 6 months to assess your progress.
As you continue to manage MND, having ongoing access to the medical team offers comfort and direction.
How Much Can Stem Cell Treatment for Motor Neuron Disease Cost?
When comparing international options, patients often observe that stem cell treatment cost differences reflect the structure of the medical program rather than just the cell product itself.
For those searching for stem cell treatment for MND in the UK or exploring options abroad, the table below provides a general overview of the global market and how programs are typically structured.
| Option | Typical Model | Broad Range | Notes |
| Stem cell treatment for MND in the UK | Private consultations and palliative focus | $15,000–$50,000+ | Limited private regenerative offerings; often requires specialized hematology units. |
| Stem cell therapy for MND in Europe | Varies by country (e.g., Germany, Switzerland) | $25,000–$55,000+ | Prices vary widely depending on the complexity of the neurodegenerative protocol. |
| Stem cell treatment for MND in the USA | Highly regulated trials or specialized private clinics | $20,000–$100,000+ | Whole-body protocols for chronic diseases typically fall in the upper range. |
| Stem cell therapy for MND at Swiss Medica | Integrated inpatient program (Diagnostics + Cells + Rehab) | €7,000–€31,000* | Comprehensive packages including accommodation, 24/7 nursing, and specialized rehabilitation. |
*Prices are indicative and based on 2026 estimates; they may vary depending on condition severity and required cell quantity.
How to Start a Consultation
The journey toward regenerative support begins with an initial consultation. During this first step, you will speak with a medical advisor to discuss your symptoms and goals.
Following the consultation, the next step is a free medical file review. Our specialists will analyze your history and medical record review. Commonly requested items include:
- EMG and MRI results
- neurology notes and diagnostic summary
- recent respiratory function tests (if available)
- current medication list and allergies
Contact us
For a structured case review, contact Swiss Medica for a consultation. The medical team can evaluate records, explain eligibility, and outline realistic supportive goals for stem cell treatment for motor neuron disease.

Medical Advisor, Swiss Medica doctor
Frequently Asked Questions
1. Can stem cells cure motor neuron disease?
No. Stem cells are discussed as supportive therapy that may help protect neural system function and increase quality of life.
2. Can stem cell therapy replace standard treatments?
No. Standard neurological management, respiratory planning, rehabilitation, and symptom control remain the foundation. Regenerative therapy is considered an adjunct when appropriate, not a replacement.
3. How long do potential effects last?
The effects of therapy peak within 3–6 months and last for a year or longer.
4. Can caregivers accompany patients?
Yes. At Swiss Medica, patients may be accompanied by a friend, family member, or caregiver. One accompanying person’s lodging and meals are included in the patient program’s cost.
5. Is Swiss Medica accessible for patients with limited mobility?
Our hospital is fully barrier-free and wheelchair accessible and designed for patients with mobility challenges.
List of References:
Ciervo Y, Ning K, Jun X, Shaw PJ, Mead RJ. Advances, challenges and future directions for stem cell therapy in amyotrophic lateral sclerosis. Mol Neurodegener. 2017 Nov 13;12(1):85. doi: 10.1186/s13024-017-0227-3.
Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, et al. (2012) Safety of Cell Therapy with Mesenchymal Stromal Cells (SafeCell): A Systematic Review and Meta-Analysis of Clinical Trials. PLOS ONE 7(10): e47559. https://doi.org/10.1371/journal.pone.0047559
Agüera-Morales E, Fernández-Sánchez VE, Navarro-Mascarell G, Cabezas-Rodríguez JA, Peña-Toledo MÁ, Reyes-Rodríguez V, Postigo-Pozo MJ, Patrignani-Ochoa G, Geniz-Clavijo MÁ, Márquez-Infante C, Tallon-Aguilar L, Tinoco-González J, Padillo-Ruiz J, Valladares-Sánchez A, Caballero-Eraso C, López-Ramírez C, Mata Alcázar-Caballero R, Leyva-Fernández L, Rodríguez-Acosta A, Maldonado-Sánchez R, García-Martín ML, Somoza-Ramírez M, Quijano-Ruiz B, Macías-Sánchez MDM, Carmona-Sánchez G, Fernández-López O, Fernández-Fernández Ó. Adipose-derived mesenchymal stem cells for the treatment of Amyotrophic Lateral Sclerosis. A phase I/II safety and efficacy clinical trial. Front Neurol. 2025 Oct 8;16:1655124. doi: 10.3389/fneur.2025.1655124.
Thonhoff JR, Ojeda L, Wu P. Stem cell-derived motor neurons: applications and challenges in amyotrophic lateral sclerosis. Curr Stem Cell Res Ther. 2009 Sep;4(3):178-99. doi: 10.2174/157488809789057392.
Juliana Ferreira Vasques, Leandro Coelho Teixeira Pinheiro, Renata Guedes de Jesus Gonçalves, Rosalia Mendez-Otero, and Fernanda Gubert. Cell-based Research and Therapy for Amyotrophic Lateral Sclerosis: Promises and Challenges. 2021 https://www.ncbi.nlm.nih.gov/books/NBK573430/
MD, Pediatrician, Regenerative Medicine Specialist